|
V) Mn is present in VII-B group and hass +7 oxidation state.įor example in case of KMnO 4 Mn shows +7 oxidation state. Iv) Cr is present in VI-B and has +6 oxidation state.įor example in case of CrO 3, H 2CrO 4, and H 2Cr 2O 7 Chromium has +6 oxidation state. Iii) vanadium is present in group V-B and shows the oxidation number of +5.įor example in case of V 2O 5 Vanadium shows +5 oxidation state. Ii) Zn, Cd, Hg are present in II-B, so their oxidation states are +2.įor example in case ZnCl 2, CdCl 2 zinc and cadmium has +2 oxidation state. In case of Cs₂CuF₆ copper has +4 oxidation state. In case of KCuO₂ and K₃CuF₆ copper has +3 oxidation state. In case of CuCl, Cu₂O, and Cu₂S copper has +1 oxidation state. The oxidation number of fluorine in all compounds is (-1). The oxidation number of hydrogen in most compounds is (+1). I) Cu, Ag, Au are present in group I-B, so their Oxidation states of Transition elements states are +1 and +3.įor example in case of CuCl 2, CuO, CuSO 4 Copper has +2 oxidation state which is most common. The oxidation number of oxygen in most compounds is (-2). They also show the oxidation state according to their group number. The oxidation number in the case of a monatomic ion is always equal to the value of the charge of. Transition elements are present in B sub-groups of the modern periodic table. The oxidation number of any free element always remains 0. The reason is that the outermost shells are completely filled and have least tendencies to gain or lose the electron. Viii) The elements of group VIII-A or zero group show zero oxidation state. 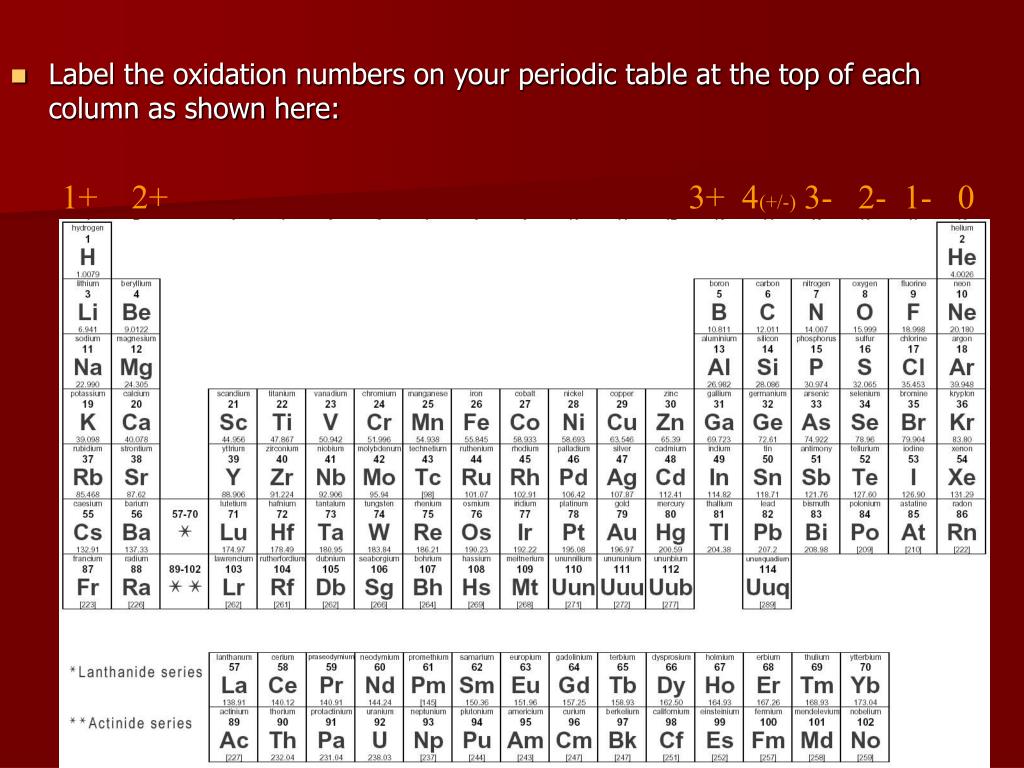
Multiple oxidation states of group VII-A elements Therefore loss of electron results into positive charge and only one electron is removed therefore, +1 is the oxidation state. Now as these are metals and lose that one electron from s-orbital. I) Elements of group I-A which have one electron in outermost shell show +1 oxidation state. Secondly see that does the atom lose electrons (metal) or gains electrons (non-metal, gases).įollowing examples will clear this concept. Its oxidation number in compounds is always -1. 'F' is the most electronegative element in the Periodic Table. These periodic tables use accurate data for name, atomic number, element symbol, atomic weight, and electron configuration, obtained from the International Union of Pure and Applied. Some tables are available as slides in Google Apps. > The most common oxidation number for Group 7 elements is -1, as in 'HF', 'CaCl'2, 'NaBr', and 'KI'. This is a collection of free printable periodic tables in PDF file or PNG image format to save, print, and use. Oxidation state rules for Calculating oxidation stateįirst of all try to know which group an atom belongs to? The oxidation number of a Group 7 element depends on the compound it is in. The number of electrons in a valance shell mostly decide about the oxidation state. The electronic distribution of elements in a particular group remains the same. Oxidation state of an atom can be predicted by the number of its valence shell electrons. The oxidation number of an atom is zero when it is present in the form of a free element. The oxidation number of Sn is +4 and that of Cl is -1. In covalent compounds the positive and negative oxidation number of an element is decided on the basis of electronegitivity of the elements. Similarly, Cl gains one electron and develops a -1 charge that becomes its valency. In this case, Na donates its one electron to chlorine and develops a +1 charge that is its balance as well. We know that metals always lose electrons and gases takes gain the electrons. It is also referred as the number of electrons an element takes or gains during a chemical reaction.įor example in case of ionic compounds such as NaCl, Na is a metal and Cl is gases. Oxidation state of an atom in a compound is the charge which it would carry in the most probable ionic formulation of ionic compound. Oxidation state rules for Calculating oxidation state.It is the 8th most abundant element in the Earth's crust, constituting 2% by mass. Antoine Bussy was the first to produce it in consistent form in 1831. 
Magnesium was first discovered in 1808 by Sir Humphry Davy in England by the electrolysis of magnesia and mercury oxide. Atomic Number = 12 Mass = 24.31 g mol -1 Electrion Configuration = 3s 2 Density = 1.738 g cm -3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
